PACE-CME
Subtitles available: 简体中, English, German & more
Optimizing treatment for patients with HoFH
Ladies and gentleman, I'm happy to share with you, the data from optimizing treatment for patients with Homozygous Familial Hypercholesterolemia. Now, let's take one recap at Homozygous Familial Hypercholesterolemia. We should know that it is the most prevalent inherited metabolic disease and compared in the graph on the right with other diseases, with inherited metabolic abnormalities. So Homozygous FH is, albeit rare, one in three hundred thousand, it's absolutely something we should take notice of.
Now, why should we take notice? I refer to my colleague Bert Wiegman, where actually you see these cases in pediatric Hypercholesterolemia. At very young ages, these children already have visible abnormalities, but the problem is, they also have paramount involvement of the coronary arteries, where you can see here a severe obstruction. And in fact in this five-year-old case, this boy died of Myocardial Infarction at the age of five.
And if we look at the recently published HICC registry data of all Homozygous Familial Hypercholesterolemia, the largest case series in the world on this disease by Tycho Tromp. You can see that the mean age of death is 32 in this cohort. So it's a horrible disease, Homozygous FH.
Now, what can we do about it? Because it used to be quite hopeless. Well, let's take one step back to the Heterozygous FH. In Heterozygous FH, we have learned that if you start treatment early between 12 to 15 and compared to the parents with heterozygous FH, that actually early treatment completely prevents events. So this is what we should actually extrapolate to the Homozygous FH: treat early and treat aggressive.
So let's see in Heterozygous how we're doing now. Look at these data. These are asymptomatic Heterozygous FH patients, who were scanned in Spain, 440, mean age 46 years old. And you can see here that more than 50% already had significant coronary disease, both with calcium and/or stenoses in the coronary arteries and that was all driven by the exposure to LDL. So early challenging the elevated Hypercholesterolemia is absolutely essential.
How are we doing in Homozygous FH? Here you can see from the HICC registry again. Less than 10% are at goal. And you see that you need an increasing number of agents accumulated. Statins, ezetimibe, PCSK9 and additional agents in order to get anywhere near the guideline. And there we're actually failing. So let me briefly go through the options of therapy in homozygous FH. One is the South African publications. Here you can see that in homozygous FH, although we know that statins and ezetimibe will not optimally work because of the defective LDL receptors. Here you can see that there is a huge impact. If you give patients nothing, before the 80s, or if you actually give them statin, albeit in combination, you can see that there's a significant improvement in outcome. We've learned from the Homozygous FH TESLA study with PCSK9 antibodies that although PCSK9s increase clearance of LDL through the liver, via the LDL receptor, you can see that still overall there's a 30% reduction in LDL by co-administration of PCSK9 inhibition in Homozygous FH.Now obviously, if you have a complete negative negative double mutation, you will not see any effect, but in all other defective mutations, you can still achieve 30%, so approximately half of the effect of in the wild types.
So what about other agents? We have seen the data from the MTP inhibitors. And the MTP inhibitors actually work on a different level. They prevent the loading of the Apo B particle both at level of the intestine, so chylomicron secretion, as well as the level of the liver. That's the VLDL secretion. So what you actually can expect, is that VLDL will be produced less, so less LDL. There's a 50% reduction, also in Homozygous FH. But obviously since unloading of the liver is impaired, you can also see that there's an accumulation of fat in the liver.
And these are elegant data by the Italian colleagues of this original study, where they show that also in the individual Italian patients, you have a marked reduction in LDL, approximately 40 to 50%. But again, in the majority of patients, you had to down titrate the MTP inhibitor because of transaminase increases. And there at the bottom you can see that the fraction of fat in the liver does increase. So this is an effective medication, but you need to be careful and exactly know what you do.
What about lipid apheresis? So if you tried everything and these severe Homozygous FH patients are still not good enough. Well here you can see the lipid apheresis efficacy. It's highly effective with an acute reduction of 60 to 80%, but the problem is after three days, you already lost 70% of your LDL-lowering capacity due to the ongoing production of LDL in the body and then only once a week apheresis. So it's effective, but unfortunately, there are no randomized controlled trials yet establishing its effect on cardiovascular disease, but it's expected in these very severe Homozygous FH that it will be beneficial.
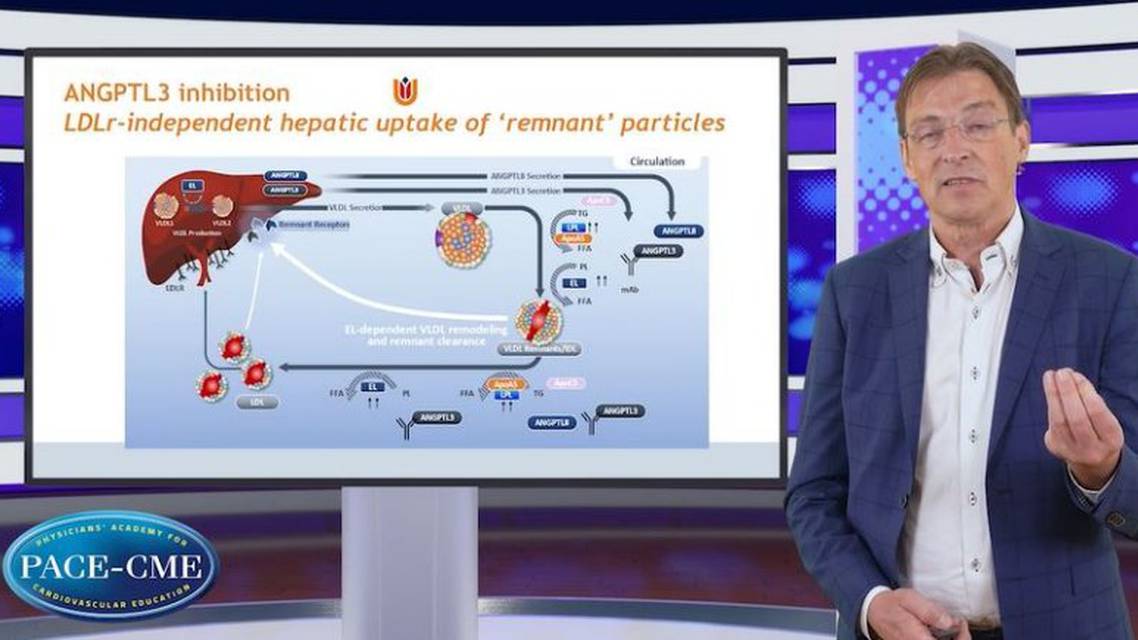
So, what about the newest kid on the block? The ANGPTL3 inhibition. So ANGPTL3 is unique because we've seen that statins and ezetimibe and the PCSK9, they all increase LDL clearance by the LDL receptor. But now the Evinacumab, which is an antibody inhibiting the ANGPTL3. What actually happens here, ANGPTL3 inhibits lipolysis, both by lipoprotein lipase and endothelial lipase. So if you use an antibody to take away the ANGPTL3 inhibition, you will actually have an increased lipolysis through both LPL and endothelial lipase. This results in a very rapid transformation of the VLDL particles into large remnant particles, and they can be taken up by the liver in an LDL receptor independent manner. And that implies that you add on top of the LDL receptor mediated clearance another independent mechanism which is LDL receptor independent. And indeed if you look at the data in Homozygous FH here you can see an approximately 50% reduction in Homozygous FH patients. That's a very big pronounced effect. And you can see that if you switch the group, they also have a 50% and look at the treatment duration. We now have treatment data for more than a year. This effect persists without any effect attenuation after prolonged therapy.
So, what about the future? Well, now we know that we have very significant options even in Homozygous FH. Or just one glance at the future is the very promising data of the CrisprCas9 gene editing. Now, we've learned from the group in the US with Sekar Kathiresan leading, we've learned that we can now edit the selected genes at the level of the liver. And look at these data in primates. In these primates you see that one single administration of the lipid formula, which includes a CrisprCas gene editing machinery, you can result in a more than 90% reduction of PCSK9 transcription in the liver. And this translates to a very prolonged reduction of LDL of more than 50% throughout many weeks of follow-up. So then maybe a single intervention could also be of value here, but obviously in Homozygous FH we might actually need more than PCSK9 because the LDL receptor is not the optimal target. Is this the far future? Well, I want to refer to my colleague, Danny Cohn who just last week actually performed the first CrisprCas9 gene therapy in Hereditary Angioedema. So this is not the far future. This is expected to be in the clinic between 2027 and 2029. So beware what's happening in CrisprCas gene editing.
So let me share you my enthusiasm because Homozygous FH used to be this horrible disease. Now look at the data from patients at our Outpatient Clinic. I just picked a couple, seven, who are being treated with statins, ezetimibe, PCSK9 inhibitor. Some of them actually are on apheresis and then in open-label extension of the evinacumab, so the ANGPTL3 antibody trial. And look at these data. I wanted to share this with you. The baseline LDLs of these patients, you can read it here, is higher than 10, some of them above 20. Now, look at the values which are just picked up out of the electronic patient file. It's 0.6, 1.1, 0.6. So these are the values. We can treat Homozygous FH,if we really try and apply the most modern treatment modalities. So we can revert this horrible outcome in this disease.
And this is just to share a little bit of my enthusiasm on imaging. These are two patients with Homozygous FH whom we actually started this four to five pronged approach for intensive LDL lowering. And we looked here at the CT angiography. And we actually observed a 60 to 80% reduction in total plaque volume when we start early. So, beautiful results give hope for the future.
So here you can see we have pre lipid-lowering therapy treatment. We have very high levels if we add all these combined interventions together we can now very effectively treat even Homozygous FH.
So as a take-home, Homozygous FH was an aggressive deadly disease of young patients. If we really perform the best interventions, we now have the capacity to completely eradicate LDL burden, even in these very severely affected Homozygous FH patients. We have to implement imaging strategies to guide our intensive treatment and make sure that we do everything to prevent this cardiovascular disease from occurring. And if we really want optimal medical care, you should know that now we need to strive for the combination of statins, ezetimibe and PCSK9, lomitapide, apheresis and potentially as soon as it has been registered, Evinacumab. Then hopefully we can also cure these very severely diseased Homozygous FH patients.
Thank you for your attention.
Recommended
- Novel LDL-c lowering therapies: What is the potential for ANGPTL3 inhibition in HoFH?Novel LDL-c lowering therapies: What is the potential for ANGPTL3 inhibition in HoFH?HoFH: The need to treat - creating awareness, understanding and insightsNovel LDL-c lowering therapies: What is the potential for ANGPTL3 inhibition in HoFH?
- HoFH: Why is early diagnosis important?HoFH: Why is early diagnosis important?HoFH: The need to treat - creating awareness, understanding and insightsHoFH: Why is early diagnosis important?





