Basal insulin and cardiovascular and other outcomes in dysglycemia
In the ORIGIN study, participants with impaired fasting glucose who were at risk for cardiovascular events were assigned to receive insulin glargine or standard care. Daily insulin glargine injections neither increased nor reduced the risks for myocardial infarction, stroke, cancer, or CV-related mortality.
Basal Insulin and Cardiovascular and Other Outcomes in DysglycemiaLiterature - The ORIGIN Trial Investigators. N Engl J Med 2012. June 9
The ORIGIN Trial Investigators
NEJM 2012 (published online before print) DOI: 10.1056/NEJMoa1203858
Background
An elevated fasting plasma glucose level is an independent risk factor for adverse cardiovascular outcomes [1-7]. Elevated fasting plasma glucose levels (above 5.6 mmol/l) indicate an insufficient endogenous insulin secretion to overcome underlying insulin resistance [8,9]. Correcting this deficiency by early provision of sufficient basal insulin to normalize fasting plasma glucose levels may reduce cardiovascular outcomes and may also reduce incident diabetes in people at risk for diabetes.
The Outcome Reduction with Initial Glargine Intervention (ORIGIN) trial tested these hypotheses.
In a 2 x 2 factorial design, more than 12,500 patients with impaired fasting glucose, impaired glucose tolerance, or diabetes were randomized to either insulin glargine or standard care.
Main results
Insulin glargine versus standard care:
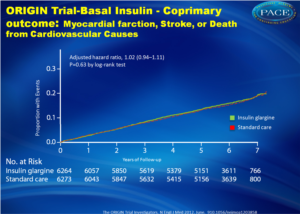
- Nonfatal MI, nonfatal stroke, or CV death: 16.6% versus 16.1% (hazard ratio, 1.02, CI 0.94 – 1.11, p = 0.63)
- Nonfatal MI, nonfatal stroke, CV death, revascularization or HF hospitalization: 28.6% versus 27.5%, HR 1.04, CI 0.97 – 1.11, p = 0.27)
Conclusion
Daily insulin glargine injections, started during the early stages of type 2 diabetes, neither increased nor reduced the risks for myocardial infarction, stroke, cancer, or CV-related mortality. The glycemic benefit of insulin glargine might have an impact on microvascular or other outcomes, but the trial does not support changing standard therapies for early dysglycemia.
References
1. Sarwar N, Gao P, Seshasai SR, et al. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: a collaborative meta-analysis of 102 prospective studies. Lancet 2010;375:2215-22.
2. Selvin E, Steffes MW, Zhu H, et al. Glycated hemoglobin, diabetes, and cardiovascular risk in nondiabetic adults. N Engl J Med 2010;362:800-11.
3. Gerstein HC, Santaguida P, Raina P, et al. Annual incidence and relative risk of diabetes in people with various categories of dysglycemia: a systematic overview and meta-analysis of prospective studies. Diabetes Res Clin Pract 2007;78:305-12.
4. Anand SS, Dagenais GR, Mohan V, et al. Glucose levels are associated with cardiovascular disease and death in an international cohort of normal glycaemic and dysglycaemic men and women: the Epi-DREAM cohort study. Eur J Cardiovasc Prev Rehabil 2011 May 6 (Epub ahead of print).
5. Gerstein HC, Islam S, Anand S, et al. Dysglycaemia and the risk of acute myocardial infarction in multiple ethnic groups: an analysis of 15,780 patients from the INTERHEART study. Diabetologia 2010;53:2509-17.
6. Seshasai SR, Kaptoge S, Thompson A, et al. Diabetes mellitus, fasting glucose, and risk of cause-specific death. N Engl J Med 2011;364:829-41. [Erratum, N Engl J Med 2011;364:1281.]
7. Turnbull FM, Abraira C, Anderson RJ, et al. Intensive glucose control and macrovascular outcomes in type 2 diabetes. Diabetologia 2009;52:2288-98. [Erratum, Diabetologia 2009;52:2470.]
8. Stumvoll M, Goldstein BJ, van Haeften TW. Pathogenesis of type 2 diabetes. Endocr Res 2007;32:19-37.
9. Turner RC, Holman RR. Insulin rather than glucose homoeostasis in the pathophysiology of diabetes. Lancet 1976;1:1272-4.
Abstract
BackgroundThe provision of sufficient basal insulin to normalize fasting plasma glucose levels may reduce cardiovascular events, but such a possibility has not been formally tested.
Methods
We randomly assigned 12,537 people (mean age, 63.5 years) with cardiovascular risk factors plus impaired fasting glucose, impaired glucose tolerance, or type 2 diabetes to receive insulin glargine (with a target fasting blood glucose level of ≤95 mg per deciliter [5.3 mmol per liter]) or standard care and to receive n–3 fatty acids or placebo with the use of a 2-by-2 factorial design. The results of the comparison between insulin glargine and standard care are reported here. The coprimary outcomes were nonfatal myocardial infarction, nonfatal stroke, or death from cardiovascular causes and these events plus revascularization or hospitalization for heart failure. Microvascular outcomes, incident diabetes, hypoglycemia, weight, and cancers were also compared between groups.
Results
The median follow-up was 6.2 years (interquartile range, 5.8 to 6.7). Rates of incident cardiovascular outcomes were similar in the insulin-glargine and standard-care groups: 2.94 and 2.85 per 100 person-years, respectively, for the first coprimary outcome (hazard ratio, 1.02; 95% confidence interval [CI], 0.94 to 1.11; P=0.63) and 5.52 and 5.28 per 100 person-years, respectively, for the second coprimary outcome (hazard ratio, 1.04; 95% CI, 0.97 to 1.11; P=0.27). New diabetes was diagnosed approximately 3 months after therapy was stopped among 30% versus 35% of 1456 participants without baseline diabetes (odds ratio, 0.80; 95% CI, 0.64 to 1.00; P=0.05). Rates of severe hypoglycemia were 1.00 versus 0.31 per 100 person-years. Median weight increased by 1.6 kg in the insulin-glargine group and fell by 0.5 kg in the standard-care group. There was no significant difference in cancers (hazard ratio, 1.00; 95% CI, 0.88 to 1.13; P=0.97).
Conclusions
When used to target normal fasting plasma glucose levels for more than 6 years, insulin glargine had a neutral effect on cardiovascular outcomes and cancers. Although it reduced new-onset diabetes, insulin glargine also increased hypoglycemia and modestly increased weight.