Persistence to medication affects clinical outcome, but the reverse is also true
Nonpersistence to medication increases the risk of CV events, but events may also lead to nonpersistence, yielding a vicious cycle with higher risk of future CV outcomes.
Effects of nonpersistence with medication on outcomes in high-risk patients with cardiovascular disease.Literature - Böhm M, Schumacher H, Laufs U et al. - Am Heart J. 2013 Aug;166(2):306-314
Böhm M, Schumacher H, Laufs U et al.
Am Heart J. 2013 Aug;166(2):306-314.e7. doi: 10.1016/j.ahj.2013.04.016.
Background
Angiotensin-converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs) provide effective secondary prevention in individuals who underwent a cardiovascular event. Nonpersistence with medications can increase the risk of CV events and death in hypertension [1-3], after acute coronary syndromes [3-6] and heart failure [7-9].The influence of CV events on persistence has not been evaluated. The Ongoing Telmisartan Alone and in combination with Ramipril Global Endpoint Trial (ONTARGET) aims to study the effects of persistence with study medication on CV outcomes in a large population of high risk patients, and in turn, whether nonfatal events affect medication persistence. 25620 Patients were randomised to telmisartan 80 mg daily, ramipril 10 mg, or the combination of both drugs, and followed up every 6 months for up to 5.5 years. Nonpersistence was defined as premature and permanent stopping of study drugs.
Main results
- 4629 patients (18.1%) stopped study medication before their final visit or death. Of these, 18.8% had previously temporarily stopped their medication. Of the 20991 persistent patients, 11.5% had temporal interruptions.
- Most discontinuations (33.7%) occurred in the first study year, and the number of patients not taking their study medication steadily increased over time.
- In a Cox model that takes into account how long a patient was on or off medication, nonpersistent patients had a significantly higher risk of CV death and chronic heart failure hospitalisation than persistent patients, but no altered risk for MI and stroke.
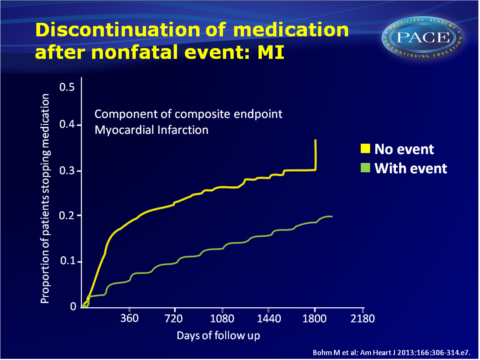
- Persistence was highest in the patients without events, while in patients who underwent an event, persistence was higher when the event occurred early, whereas persistence was better when they experienced the event at a later stage. An increasing number of events further negatively affected persistence.
- Patients who were nonpersistent after a primary event experienced more subsequent events (adjusted HRs vs. persistent patients: 1.54, 95%CI: 1.13-2.11, P=0.0004) than patients who became nonpersistent before an event (adjHR vs. persistent) (HR: 1.31, 95%CI: 1.16-1.48, P<0.0001).
- Of the nonpersistent patients, 54% of study medication discontinuation could be explained to adverse events. In patients who stopped medication before a primary event, no difference was seen between whether patients stopped due to an adverse events or another reason.
Download PACE Bohm AHA 2013.pptx

Conclusion
Patients who were nonpersistent to protective therapies showed higher CV event rates. Events may however not only occur as a result of nonpersistence, but nonpersistence also seemed to result from a previous nonfatal event. A vicious cycle develops yielding higher risk for future CV outcomes. Identification of reduced persistence in these high-risk groups might help reduce CV events.References
1. Mazzaglia G, Ambrosioni E, Alacqua M, et al. Persistence to antihypertensive medications and cardiovascular morbidity among newly diagnosed hypertensive patients. Circulation 2009;120: 1598-605.
2. Chobanian A. Impact of nonpersistence to antihypertensive therapy. Circulation 2009;120:1558-60.
3. Simspon S, Eurich DT, Majumdar SR, et al. A meta-analysis of the association between persistence to drug therapy and mortality. Br Med J 2006;333:15.
4. Coronary Drug Project Research Group. Influence of persistence to treatment and response of cholesterol on mortality in the coronary drug project. N Engl J Med 1980;303:1038-41.
5. Horwitz RI, Viscoli CM, Berkman L, et al. Treatment persistence and risk of death after a myocardial infarction. Lancet 1990;336:542-5.
6. Rasmussen J, Chong A, Alter D. Relationship between persistence to evidence-based pharmacotherapy and long-term mortality after acute myocardial infarction. JAMA 2007;297:177-86.
7. Bennett SJ, Huster GA, Baker SL, et al. Characterization of the precipitants of hospitalization for heart failure decompensation. Am J Crit Care 1998;7:168-74.
8. Ghali JK, Kadakia S, Cooper R, et al. Precipitating factors leading to decompensation of heart failure: traits among urban blacks. Arch Intern Med 1988;148:2013-6.
9.Granger BB, Swedberg K, Ekman I, et al, CHARM investigators. Persistence to candesartan and placebo and outcomes in chronic heart failure in the CHARM programme: double-blind, randomized, controlled clinical trial. Lancet 2005;366:2005-11.
Find this article on Pubmed
