Post-MI patients with multi-vessel disease may benefit from long-term P2Y12 inhibitor therapy
Patients with a history of MI and MVD are at increased risk for MACE and coronary events compared to those without MVD and benefit from long-term ticagrelor therapy.
Ticagrelor for Secondary Prevention of Atherothrombotic Events in Patients With Multivessel Coronary DiseaseLiterature - Bansilal S, Bonaca MP, Cornel JH, et al. - J Am Coll Cardiol 2018;71:489–96
Background
In the PEGASUS-TIMI 54 (Prevention of Cardiovascular Events in Patients With Prior Heart Attack Using Ticagrelor Compared to Placebo on a Background of Aspirin–Thrombolysis In Myocardial Infarction 54) trial, ticagrelor reduced the risk of MACE (CV death, MI, or stroke) and increased non-fatal major bleeding in high risk patients with a history of MI [1,2].
In this pre-specified analysis from the PEGASUS-TIMI 54 trial, the efficacy and safety of long-term ticagrelor therapy was evaluated in patients with a history of MI and multi-vessel coronary artery disease (MVD).
In the PEGASUS-TIMI 54 trial, patients with a history of MI 1 to 3 years previously and ≥1 additional CV risk factor (age >65 years, diabetes mellitus requiring medication, second prior spontaneous MI, chronic renal dysfunction, or MVD) were randomized to receive ticagrelor (90 mg or 60 mg twice daily) or placebo, on top of low-dose aspirin, and followed for a median of 33 months.
For the present analysis, MVD was defined the presence of >50% stenosis in ≥2 separate major coronary territories at the time of the index event (with or without prior revascularization).The primary efficacy endpoint was MACE, a composite of CV death, MI, or stroke, and the primary safety endpoint was Thrombolysis In Myocardial Infarction (TIMI) major bleeding. An additional efficacy endpoint was the composite of coronary death, MI, or stent thrombosis, and intracranial hemorrhage (ICH), and fatal bleeding.
Main results
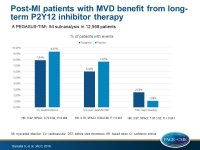
- Of 21,162 patients randomized in the PEGASUS-TIMI 54 trial, 12,558 (59.4%) had a history of MVD.
- Compared with individuals with single-vessel disease, patients with MVD were at higher risk for MACE (9.37% vs. 8.57%; HRadj: 1.24; 95%CI: 1.03-1.50; P=0.026) and coronary events (7.67% vs. 5.34%; HRadj: 1.49; 95%CI: 1.19-1.87; P=0.0005).
- In MVD patients, compared with placebo, ticagrelor led to significant reductions of MACE (pooled data from the 2 doses tested: 7.94% vs. 9.37%; HR: 0.82; 95%CI: 0.72-0.94; P=0.004), coronary events (6.02% vs. 7.67%; HR: 0.76; 95%CI: 0.66-0.88; P < 0.001), a 36% reduction in coronary death (HR: 0.64; 95%CI: 0.48-0.85; P=0.002), a 21% reduction in MI (HR: 0.79; 95% CI: 0.67 to 0.93), and a 41% reduction in stent thrombosis (HR: 0.59; 95% CI: 0.37 to 0.94).
- Ticagrelor increased the risk of TIMI major bleeding (2.52% vs. 1.08%, HR: 2.67; 95%CI: 1.81-3.93; P<0.001), but not ICH or fatal bleedings.
Conclusion
Patients with a history of MI and MVD had an increased risk for MACE and coronary events compared to those with single-vessel disease. Long-term ticagrelor therapy, on top of aspirin, reduced substantially the risk of MACE in patients with prior MI and MVD and increased the risk of TIMI major bleeding, but not ICH or fatal bleeding. It will be interesting to study the safety and efficacy of ticagrelor monotherapy compared to dual antiplatelet therapy.
Editorial comment
In their editorial article, Lee and Tanoue [3] review data related to the debate regarding the optimal duration of dual antiplatelet therapy (DAPT) post-PCI, and note that the results reported by Bansilal et al. support a higher intensity and extended duration of antiplatelet therapy in post-MI patients with multi-vessel coronary artery disease. The authors are intrigued by this approach of higher intensity and extended duration of DAPT as opposed to revisiting revascularization strategies to manage MVD patients. Although the pathophysiology mechanism for the increased risk of coronary events and death post-MI in MVD patients is unknown, it is clear that these patients benefit from ticagrelor therapy. The authors conclude: ‘Taken together, the PEGASUS-TIMI 54 subset analyses give validation to existing data supporting improved outcomes with higher intensity and longer duration of antiplatelet therapy with ticagrelor in certain high-risk patient cohorts. Specifically, the data support that the presence of MVD may be an indication to pursue extended DAPT with ticagrelor for up to 3 years in the absence of limiting adverse effects.’
References
1. Bonaca MP, Bhatt DL, Braunwald E, et al. Design and rationale for the Prevention of Cardiovascular Events in Patients With Prior Heart Attack Using Ticagrelor Compared to Placebo on a Background of Aspirin-Thrombolysis in Myocardial Infarction 54 (PEGASUS-TIMI 54) trial. Am Heart J 2014;167:437–44.e5.
2. Bonaca MP, Bhatt DL, Cohen M, et al. Long-term use of ticagrelor in patients with prior myocardial infarction. N Engl J Med 2015;372:1791–800.
3. Michael S. Lee, MD, Michael T. Tanoue, MD. Impact of Ticagrelor in Patients With Prior MI and MVD for Reducing Cardiovascular Events. J Am Coll Cardiol 2018;71:497-8.