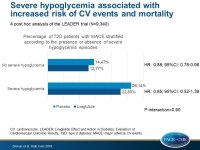
Severe hypoglycemia associated with increased risk of CV events and mortality
In the LEADER trial, patients with severe hypoglycemia episodes had a higher risk of CV events and mortality compared with patients without severe hypoglycemia, independent of treatment group.
Hypoglycemia, Cardiovascular Outcomes, and Death: The LEADER ExperienceLiterature - Zinman B, Marso SP, Christiansen E, et al. - Diabetes Care 2018; published online ahead of print
Introduction and Methods
There is an association between hypoglycemia and an increased risk of CV events and mortality, although the evidence for a causal effect is limited [1-3]. In this post hoc analysis of the LEADER (Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results) trial [4], the associations between hypoglycemia and CV outcomes and mortality were investigated in patients with type 2 diabetes (T2DM).
In the double-blind, randomized, placebo-controlled LEADER trial, the glucagon-like peptide 1 analog liraglutide significantly reduced CV events and mortality compared with placebo, in T2DM patients at high CV risk, who additionally received standard care, and were followed for 3.5-5 years.
The primary end point in LEADER was the time to first occurrence of a major adverse CV event (MACE), including CV death, non-fatal myocardial infarction, or non-fatal stroke. Hypoglycemia was a secondary self-reported safety endpoint, and severe hypoglycemia was defined according to the American Diabetes Association criteria [5].
Main results
- Out of a total of 9,340 T2DM patients, 45% had a confirmed episode of hypoglycemia, and 3% had at least one severe hypoglycemia event.
- Severe and confirmed hypoglycemia rates were higher in patients receiving insulin at baseline, and significantly lower in the liraglutide group compared with the placebo group.
- Patients with severe hypoglycemia had a significant higher risk of MACE (HR: 1.9, 95%CI: 1.5-2.5, P<0.0001), CV death (HR: 2.2, 95%CI;1.5-3.2, P<0.0001), non-CV death (HR: 2.3, 95%CI;1.4-3.6, P<0.001) and all-cause death (HR: 2.2, 95%CI;1.7-3.0, P<0.0001) compared with those who did not have severe hypoglycemia episodes.
- The risk of MACE, CV death, non-CV death, or all-cause death was 7- to 15-fold higher in the first 7 days after a severe hypoglycemia episode.
- The effect of liraglutide on risk of first MACE was similar in patients with (HR: 0.85; 95%CI: 0.52-1.39) or without (HR: 0.88; 95%CI: 0.78-0.98) severe hypoglycemia (P-interaction=0.90).
- Patients with a confirmed hypoglycemia episode had a higher risk of non-CV death compared with those who did not (HR: 1.40; 95%CI: 1.12-1.74), but there was no significant difference in the risk for MACE (HR: 1.07; 95%CI: 0.95-1.20), CV death (HR: 0.98; 95%CI: 0.82-1.18), or all-cause death (HR: 1.13; 95%CI: 0.98-1.31).
- Each confirmed hypoglycemia episode significantly increased the subsequent risk of all-cause death (HR: 1.01; 95%CI: 1.00-1.02) and severe hypoglycemia (HR: 1.03; 95%CI: 1.02-1.03).
Conclusion
In the LEADER trial, patients with severe hypoglycemia episodes had a higher risk of MACE, CV death, non-CV death, and all-cause death compared with patients without severe hypoglycemia, particularly in the first 7 days after the hypoglycemic episode. These associations were independent of treatment group.
References
1. Goto A, Arah OA, Goto M, et al. Severe hypoglycaemia and cardiovascular disease: systematic review and meta-analysis with bias analysis. BMJ 2013;347:f4533
2. Khunti K, Davies M, Majeed A, et al. Hypoglycemia and risk of cardiovascular disease and all-cause mortality in insulin-treated people with type 1 and type 2 diabetes: a cohort study. Diabetes Care 2015;38:316–322
3. Hanefeld M, Frier BM, Pistrosch F. Hypoglycemia and cardiovascular risk: is there a major link? Diabetes Care 2016;39(Suppl. 2):S205–S209
4. Marso SP, Daniels GH, Brown-Frandsen K, et al.; LEADER Steering Committee; LEADER Trial Investigators. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2016;375:311–322
5. American Diabetes Association Workgroup on Hypoglycemia. Defining and reporting hypoglycaemia in diabetes: a report from the American Diabetes Association Workgroup on Hypoglycemia. Diabetes Care 2005;28:1245–1249