GLP-1 receptor agonist therapy associated with decreased CV risk in T2DM patients with CKD
In a posthoc analysis of the LEADER trial, liraglutide treatment was associated with fewer CV outcomes patients with type 2 diabetes and chronic kidney disease at high CV risk.
Effects of Liraglutide versus Placebo on Cardiovascular Events in Patients with Type 2 Diabetes and Chronic Kidney Disease: Results from the LEADER TrialLiterature - Mann JFE, Fonseca V, Mosenzon O, et al. - Circulation 2018; published online ahead of print
Introduction and methods
Patients with type 2 diabetes (T2D) and chronic kidney disease (CKD) have an increased cardiovascular (CV) risk. Furthermore, low estimated glomerular filtration rate (eGFR) and albuminuria are independent predictors of CV outcomes [1,2]. However, it is unclear whether therapeutic interventions to decrease CV risk improve outcomes in these patients. In the Liraglutide Effect and Action in Diabetes: Evaluation of CV outcome Results (LEADER) trial [3,4], liraglutide therapy was associated with a reduced risk of CV outcomes, all-cause death and renal outcomes compared with placebo, in T2D patients with established CV disease on standard of care.
LEADER was a multicenter, double-blind, placebo-controlled trial, which randomized T2D patients at high CV risk to receive 1:1 liraglutide (1.8 mg daily or maximum tolerated dose) or placebo, on top of standard of care. The study was designed to recruit a subgroup of at least 660 patients with an eGFR
<60 mL/min/1.73 m2, approximately 220 patients with severe renal impairment (eGFR <30 mL/min/1.73 m2), and at least 440 patients with moderate renal impairment (CKD stage 3; eGFR 30–60 mL/min/1.73 m2).
This posthoc analysis assessed the effect of liraglutide versus placebo on CV and safety outcomes in subgroups of patients with eGFR <60 mL/min/1.73 m2 versus eGFR ≥60 mL/min/1.73 m2, and those with micro/macroalbuminuria (<30 mg/g versus ≥30 mg/g creatinine), versus those without albuminuria at randomization. The primary outcome was a composite of CV death, non-fatal myocardial infarction (MI), or non-fatal stroke.
Main results
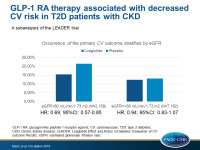
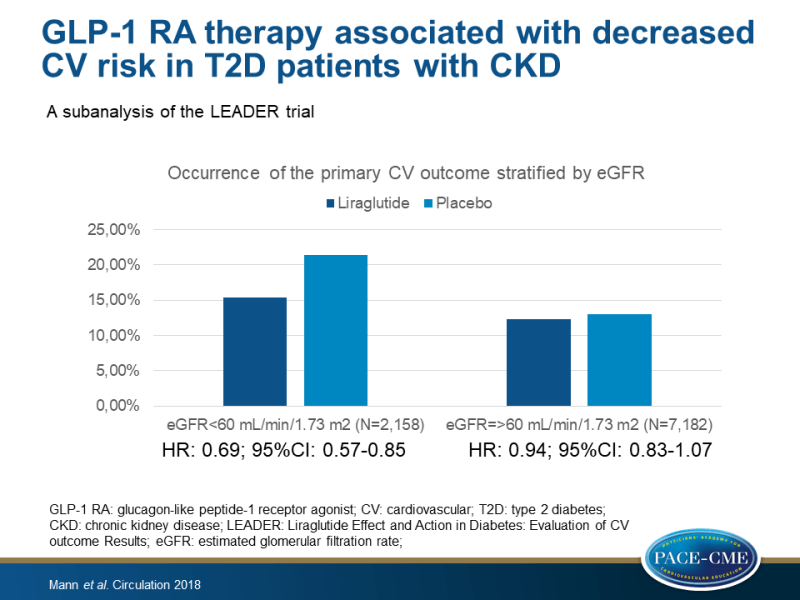
- Overall, 2,158 patients had baseline eGFR <60, and 7,182 patients had baseline eGFR >60 mL/min/1.73 m2.
- The primary composite CV outcome occurred in a lower proportion of patients taking liraglutide compared with placebo (15.4% vs 21.4% in those with eGFR <60 mL/min/1.73 m2, and 12.3% vs 13.0% in those with eGFR ≥60 mL/min/1.73 m2).
- For the primary composite outcome, the risk reduction with liraglutide was greater in the subgroup of patients with eGFR <60 mL/min/1.73 m2 (HR: 0.69; 95%CI: 0.57-0.85) than in those with eGFR ≥60 mL/min/1.73 m2 (HR: 0.94; 95%CI: 0.83-1.07; interaction P=0.01).
- When eGFR was grouped in intervals of 15 ml/min/1.73 m2, there was no consistent difference in the treatment effect of liraglutide on the primary outcome, across eGFR subgroups (interaction P=0.13). Nor was an interaction seen between eGFR at baseline and the treatment effect, with eGFR as a continuous variable.
- The HRs for the primary and key secondary outcomes for the micro/macroalbuminuria and normoalbuminuria subgroups by baseline eGFR and time to primary composite CV outcome in patients according to baseline eGFR were 0.83 (95%CI: 0.71-0.97) and 0.92 (95%CI: 0.79-1.07; P-interaction=0.36), respectively.
Conclusion
The authors conclude that in patients with type 2 diabetes and chronic kidney disease at high cardiovascular risk, liraglutide treatment was associated with decreased CV outcomes, similarly to patients without chronic kidney disease.
References
1. Mann JF, Gerstein HC, Pogue J, et al. Renal insufficiency as a predictor of cardiovascular outcomes and the impact of ramipril: the HOPE randomized trial. Ann Intern Med. 2001;134:629–636.
2. Gerstein HC, Mann JF, Yi Q, et al; HOPE Study Investigators. Albuminuria and risk of cardiovascular events, death, and heart failure in diabetic and nondiabetic individuals. JAMA. 2001;286:421–426.
3. Marso SP, Daniels GH, Brown-Frandsen K, et al; LEADER Steering Committee; LEADER Trial Investigators. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375:311–322.
4. Mann JFE, Ørsted DD, Brown-Frandsen K, et al; LEADER Steering Committee and Investigators. Liraglutide and renal outcomes in type 2 diabetes. N Engl J Med. 2017;377:839–848.