No mortality benefit of beta-blocker in stable CAD beyond one year after MI, according to registry data
CLARIFY registry data suggest that beta-blocker use was only associated with lower 5-year mortality in patients enrolled in the year following in MI. Use of calcium antagonists did not impact mortality.
β-blockers, calcium antagonists, and mortality in stable coronary artery disease: an international cohort studyLiterature - Sorbets E, Steg PG, Young R et al., - Eur Heart J. 2019:40(18):1399-1407
Introduction and methods
Beta-blockers and calcium antagonists are recommended for treatment of angina in patients with stable coronary artery disease (CAD) and widely used in patients with stable CAD, also in patients without angina. It is, however, uncertain how effective they are in improving symptoms, although in observational studies associations were noted between beta-blocker use and improved outcomes in acute coronary syndromes [1-3]. No RCTs have investigated the efficacy of beta-blockers compared with placebo in stable CAD [4,5]. Some trials have suggested a benefit of calcium antagonists on CV morbidity in stable CAD, but no data support a benefit on mortality [6,7]. In comparisons of long-acting dihydropyridines or non-dihydropyridines with beta-blockers in stable CAD, no difference was seen in their effects on clinical outcomes [8-11].
This study used data of the prospective observational longitudinal CLARIFY registry [12] of patients with stable CAD, to assess the association between beta-blocker or calcium antagonist use and clinical outcomes, with a focus on mortality. The CLARIFY registry enrolled 32703 stable CAD outpatients in 45 countries between November 2009 and June 2010, who were followed annually for up to 5 years. Patients had MI or CABG or PCI at least 3 months ago, chest pain with proven myocardial ischemia or previous coronary angiography with ≥1 coronary stenosis >50%. Complete data were available for 22006 participants who used beta-blockers and for 22004 who used calcium antagonists.
The authors acknowledge that this is a post-hoc analysis, meaning that analyses were not prespecified, and that residual confounding factors can persist even after adjustment.
Main results
Beta-blockers
- At baseline, 17135 (77.9%) of patients used beta-blockers. In patients for whom dose was available, 45.1% received less than half of the target dose, 41.6% half to less than full dose, and 13.3% received the full target dose.
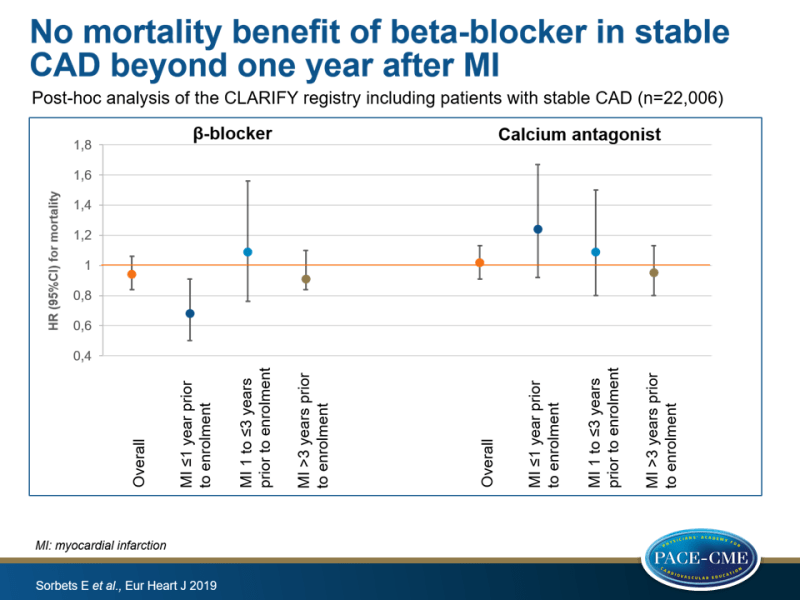
- Overall, mortality rate was 1.80 per 100 patient-years (PY). Beta-blocker use was not associated with a significant reduction of all-cause mortality (HR: 0.94, 95%cI: 0.84-1.06), CV mortality (HR: 0.91, 95%CI: 0.79-1.05) and the composite of CV mortality/non-fatal MI (HR: 1.03, 95%CI: 0.91-1.16).
- A difference was seen in effect of beta-blocker use based on time elapsed since MI. In patients with MI ≤1 year prior to enrolment, beta-blocker use was associated with a significantly lower risk of all-cause mortality (7.0% vs. 10.3%, HR: 0.68, 95%CI: 0.50-0.91), CV mortality (4.5% vs. 8.5%, HR: 0.52, 95%CI: 0.37-0.73) and CV mortality/non-fatal MI (7.2% vs. 10.3%, HR: 0.69, 95%CI: 0.52-0.93). In patients in whom MI occurred over a year ago (analyzed as 1 to ≤3 years and >3 years prior to enrolment) beta-blocker use did not significantly impact outcomes.
- In those with no history of PCI at baseline, beta-blocker use at baseline was associated with lower CV death (6.1% vs. 7.4%, HR: 0.81, 95%CI: 0.67-0.98). When categorizing by presence of angina, no association was seen between beta-blocker use and mortality.
Calcium antagonists
- At baseline, 5885 (26.7%) of patients used calcium antagonists. 79.8% of those received long-acting dihydropyridines, 14.7% diltiazem and 4.9% verapamil.
- No association was found between calcium antagonist use and mortality (HR: 1.02, 95%CI: 0.91-1.13), irrespective of having undergone PCI or presence of angina. None of the groups based on time since MI showed a significant benefit of calcium antagonist use on all-cause mortality.
Conclusion
This analysis of an international registry of contemporary stable CAD patients with a high rate use of evidence-based secondary prevention therapies, showed that beta-blocker use at baseline was associated with lower 5-year all-cause mortality only in patients enrolled in the first year following MI. Use of calcium antagonists did not impact outcomes in this patient group.
Editorial comment
In their editorial comment, Nissen and Reed [13] discuss the value of different types of data sources, and what may or may not be concluded based on the data obtained. Observational studies may be useful in generating hypotheses, which should then be proven by conducting RCTs. Moreover, they may be useful to study a question or patient population in which it is not feasible that an RCT will be conducted. Well-designed observational studies should have a narrow hypothesis, with a formally pre-specified statistical plan, ideally on prospectively collected data. Data should be robustly adjusted for potential confounders to correct for overt bias and sensitivity analyses should be performed to correct for hidden bias.
About the study by Sorbets et al., Nissen and Reed note that some of the characteristics of good observational study design were applied, but many potential confounding variables were not reported, such as socioeconomic status, frailty indices, BMI and follow-up lipid levels. Moreover, the collected variables were not balanced between patients receiving the treatment of interest and others. This is relevant in light of the concept of confounding by indication. In addition, the unblinded nature and the fact that primary endpoint and method of analysis were not prespecified, may represent other sources of bias.
Nissen and Reed applaud the authors for acknowledging nearly all of these concerns in their manuscript. ‘Their cautionary conclusions and the tone of the manuscript are a credit to the authors’ commitment to scientific accuracy and integrity. Although the conclusions of this study may ultimately prove correct, the reliability of the findings is too limited to apply this study to clinical decision-making.’
References
1. D’Ascenzo F, Celentani D, Brustio A, et al. Association of beta-blockers with survival on patients presenting with . ACS treated with PCI: a propensity score analysis from the BleeMACS registry. Am J Cardiovasc Drugs 2018;18:299–309.
2. Dondo TB, Hall M, West RM, et al. Beta-blockers and mortality after acute myocardial infarction in patients without heart failure orventricular dysfunction. J Am Coll Cardiol 2017;69:2710–2720.
3. Goldberger JJ, Bonow RO, Cuffe M, et al; OBTAIN Investigators. Effect of beta-blocker dose on survival after acute myocardial infarction. J Am Coll Cardiol 2015;66:1431–1441.
4. Freemantle N, Cleland J, Young P, et al. b Blockade after myocardial infarction: systematic review and meta regression analysis. BMJ 1999;318: 1730–1737.
5. Bangalore S, Makani H, Radford M, et al. Clinical outcomes with beta-blockers for myocardial infarction: a meta-analysis of randomized trials. Am J Med 2014: 127: 939-953
6. Poole-Wilson PA, Lubsen J, Kirwan BA, et al; Coronary disease Trial Investigating Outcome with Nifedipine gastrointestinal therapeutic system investigators. Effect of long-acting nifedipine on mortality and cardiovascular morbidity in patients with stable angina requiring treatment (ACTION trial): randomised controlled trial. Lancet 2004;364:849–857.
7. Nissen SE, Tuzcu EM, Libby P, et al. Investigators C. Effect of antihypertensive agents on cardiovascular events in patients with coronary disease and normal blood pres- sure: the CAMELOT study: a randomized controlled trial. JAMA 2004;292: 2217–2225.
8. Dargie HJ, Ford I, Fox KM. Total ischaemic burden European Trial (TIBET). Effects of ischaemia and treatment with atenolol, nifedipine SR and their combin- ation on outcome in patients with chronic stable angina. The TIBET Study Group. Eur Heart J 1996;17:104–112.
9. Rehnqvist N, Hjemdahl P, Billing E, et al. Effects of metoprolol vs verapamil in patients with stable angina pectoris. The Angina Prognosis Study in Stockholm (APSIS). Eur Heart J 1996;17:76–81.
10. Pepine CJ, Handberg EM, Cooper-DeHoff RM, et al. A calcium antagonist vs a non-calcium antagonist hypertension treatment strategy for patients with cor- onary artery disease. The International Verapamil-Trandolapril Study (INVEST): a randomized controlled trial. JAMA 2003;290:2805–2816.
11. Bangalore S, Parkar S, Messerli FH. Long-acting calcium antagonists in patients with coronary artery disease: a meta-analysis. Am J Med 2009;122:356–365.
12. Sorbets E, Greenlaw N, Ferrari R, Ford I, Fox KM, Tardif J-C, Tendera M, Steg . PG. Rationale, design, and baseline characteristics of the CLARIFY registry of . outpatients with stable coronary artery disease. Clin Cardiol 2017;40:797–806.
13. Nissen SE and Reed GW. Can we trust observational data for clinical decision-making? Eur. Heart J. 2018. doi:10.1093/eurheartj/ehy874
