Hypertriglyceridemia is not always adequately managed with statin monotherapy
A substantial number of patients with hypertriglyceridemia do not reach the recommended TG levels with statin monotherapy, and need additional TG-lowering therapy.
A VOYAGER Meta-Analysis of the Impact of Statin Therapy on Low-Density Lipoprotein Cholesterol and Triglyceride Levels in Patients with HypertriglyceridaemiaLiterature - Karlson BW, et al. Am J Cardiol 2016
Karlson BW, Palmer MK, Nicholls SJ, et al.
The American Journal of Cardiology 2016;published online ahead of print
Background
Elevated plasma triglyceride levels have been shown to be independent predictors of cardiovascular risk, even in patients who have achieved their LDL-C treatment goals with statin therapy [1,2]. LDL-C reduction with statins remains the primary treatment goal for these patients, although previous analyses have found no clear relationship between statin-mediated reductions in LDL-C and reductions in TG [3].For severe hypertriglyceridemia, defined as a TG level of >885 mg/dL [>10.0 mmol/L]), which is also a risk factor for pancreatitis, clear management guidance exists, using agents that target mainly elevated TG levels [3,4]. However, it is not very clear how to treat mild to moderate hypertriglyceridemia (defined as a TG level of 177–885 mg/dL [2.0–10.0 mmol/L]) for which, according to guidelines, a TG level of <150 mg/dL (<1.7 mmol/L) is desirable [5,6].
In this analysis, LDL-C and TG reductions were evaluated in 15,800 patients with TG ≥177 mg/dL (≥2.0 mmol/L) who were receiving treatment with different statins and doses in randomised direct comparison studies. The source of patient data is the indiVidual patient meta-analysis Of statin therapY in At risk Groups: Effects of Rosuvastatin, atorvastatin and simvastatin (VOYAGER) database [7].
Main results
• Reductions in LDL-C: There was a clear relationship between increasing statin dose and reduction in LDL-C. Across all statins and doses, the mean percent reduction (±SEM) in LDL-C ranged from –26.9% (2.0%) to –55.5% (0.8%)- the LDL-C reductions with rosuvastatin 10, 20, and 40 mg were significantly greater than those with equal or double mg doses of atorvastatin and simvastatin (P<0.05 for all comparisons)
- the LDL-C reductions in with atorvastatin 80 mg were significantly greater than those with rosuvastatin 5 and 10 mg (P<0.05)
- the LDL-C reductions with rosuvastatin 5, 10 and 20 mg were greater than those with simvastatin 20, 40 and 80 mg, respectively (P<0.05)
- rosuvastatin 10 mg resulted in a significantly greater reduction in TG compared with atorvastatin 10 mg (P<0.001)
- TG reductions with rosuvastatin 20 and 40 mg were similar to those observed with equal doses of atorvastatin
- atorvastatin 80 mg resulted in significantly greater reductions in TG than rosuvastatin 10 mg (P<0.05)
- rosuvastatin 10–40 mg resulted in significantly greater TG reductions compared with equal or double doses of simvastatin (P<0.05)
- for atorvastatin 10, 20, 40, and 80 mg: 30.3%, 38.5%, 44.8%, and 54.8% respectively
- for rosuvastatin 5, 10, 20, and 40 mg: 28.3%, 35.0%, 38.3%, and 48.9% respectively
- for simvastatin 10, 20, 40, and 80 mg: 22.6%, 25.4%, 25.9%, and 35.7% respectively
Download Karlson AJC 2016.pptx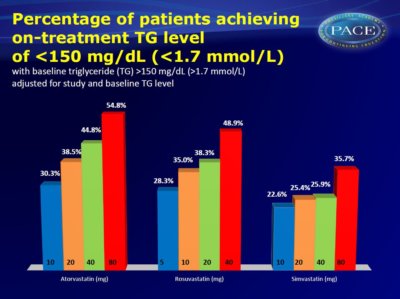
Conclusion
In patients with hypertriglyceridemia, statin monotherapy resulted in TG reductions, however, a substantial number of patients did not achieve the recommended TG level of <150 mg/dL (<1.7 mmol/L). Additional TG-lowering therapy should be considered to further reduce the residual cardiovascular risk in patients with persisting high TG levels, even if LDL-C goals are met with statin therapy.Find this article online
References
1. Nordestgaard BG, Varbo A. Triglycerides and cardiovascular disease. Lancet 2014;384:626–635
2. Miller M, Cannon CP, Murphy SA, et al. Impact of triglyceride levels beyond low-density lipoprotein cholesterol after acute coronary syndrome in the PROVE IT-TIMI 22 trial. J Am Coll Cardiol 2008;51:724–730.
3. Berglund L, Brunzell JD, Goldberg AC, et al. Evaluation and treatment of hypertriglyceridemia: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 2012;97:2969–2989
4. Hegele RA, Ginsberg HN, Chapman MJ, et al. The polygenic nature of hypertriglyceridaemia: implications for definition, diagnosis, and management. Lancet Diabetes Endocrinol 2014;2:655–666
5. Catapano AL, Reiner Z, De Backer G, et al. ESC/EAS guidelines for the management of dyslipidaemias. The Task Force for the Management of Dyslipidaemias of the European Society of Cardiology (ESC) and the European Atherosclerosis Society (EAS). Atherosclerosis 2011;217:3–46
6. Chapman MJ, Ginsberg HN, Amarenco P, et al. Triglyceride-rich lipoproteins and high-density lipoprotein cholesterol in patients at high risk of cardiovascular disease: evidence and guidance for management. Eur Heart J 2011;32:1345–1361
7. Nicholls SJ, Brandrup-Wognsen G, Palmer M, et al. Meta-analysis of comparative efficacy of increasing dose of atorvastatin versus rosuvastatin versus simvastatin on lowering levels of atherogenic lipids (from VOYAGER). Am J Cardiol 2010;105:69–76
